Kingst Medical Multi-Gas Analysis and EtCO₂ Products Receive EU MDR CE Certification
In 2026, ekingst Medical announced that its Multi-Gas Analysis Series and End-Tidal Carbon Dioxide (EtCO₂) Series products have officially obtained EU Medical Device Regulation (EU) 2017/745 (MDR) CE certification (Certificate No.: MDR 798520). This milestone is a powerful testament to ekingst's technical expertise in respiratory gas monitoring.
.png)
1. Respiratory Gas Monitoring: The "Eyes" of Clinical Safety
From the operating room to the ICU, from emergency medicine to sleep medicine — in these high-stakes clinical environments, healthcare professionals cannot assess a patient's ventilation status through sensation alone. Respiratory gas monitoring devices serve as clinicians' indispensable "eyes."
End-tidal carbon dioxide (EtCO₂) and multi-gas analysis are two core technologies in this field. EtCO₂ focuses on continuous tracking of ventilation function, while multi-gas analysis simultaneously delivers concentration data for multiple gas species. Together, they form a comprehensive respiratory monitoring solution covering every clinical scenario.

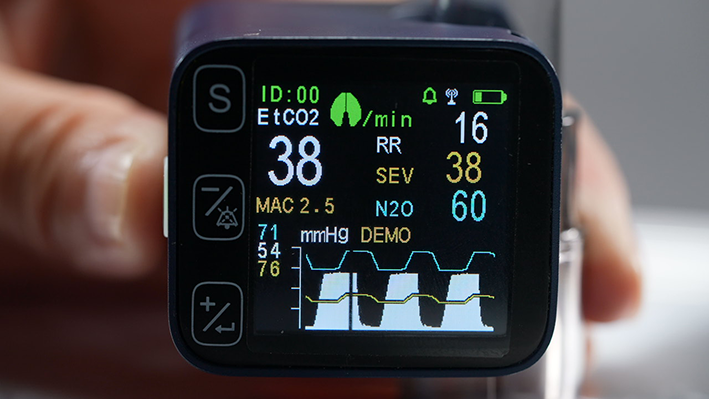
2. Multi-Gas Analysis Series: Precision Capture of Every Breath
Multi-Gas Analysis technology uses infrared absorption principles to perform real-time, quantitative analysis of CO₂, O₂, N₂O, and inhaled anesthetic agents (such as sevoflurane, desflurane, and isoflurane) in a patient's exhaled breath. With each respiratory cycle, the system simultaneously outputs inspiratory concentration (Fi), end-tidal concentration (Et), and gas waveform curves.
Core Technical Highlights:
Simultaneous Multi-Gas Monitoring: A single device can analyze CO₂, O₂, N₂O, and multiple anesthetic agents without switching probes or modules, simplifying clinical workflow and reducing equipment costs.
High-Precision Infrared Sensing: Based on infrared spectral absorption, the system delivers accurate gas identification with rapid response, ensuring data authentically reflects the patient's respiratory circuit.
Automatic Anesthetic Gas Recognition: The device auto-identifies and labels anesthetic gas types, preventing monitoring errors from manual selection and enhancing clinical safety.
Real-Time Waveform + Numerical Dual Display: Outputs gas concentration values and continuous capnogram curves simultaneously, enabling clinicians to assess patient status from both trend and morphological perspectives.
Typical Application Scenarios:
General Anesthesia Surgery: Real-time anesthetic gas concentration monitoring assists anesthesiologists in assessing depth, ensuring patients remain safely under adequate analgesia, sedation, and muscle relaxation — while helping prevent intraoperative awareness or excessive anesthesia.
Intensive Care: For ICU patients on mechanical ventilation, continuous tracking of CO₂ and O₂ changes supports lung-protective ventilation strategies that help reduce the risk of ventilator-induced lung injury (VILI).
Post-Anesthesia Care Unit (PACU): During extubation and emergence, EtCO₂ monitoring ensures airway patency and enables early detection of respiratory depression.
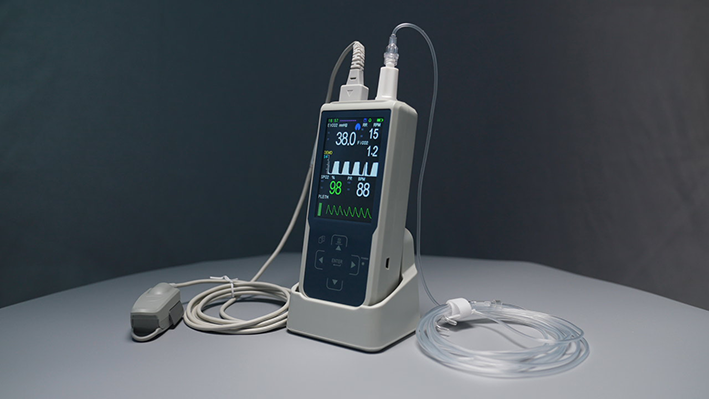
3. End-Tidal Carbon Dioxide (EtCO₂) Series: The Gold Standard in Ventilation Monitoring
EtCO₂ monitoring is one of the most reliable non-invasive methods for assessing ventilation function. By continuously measuring end-tidal CO₂ partial pressure and respiratory rate with each breath, it provides real-time insight into a patient's ventilation efficiency, pulmonary circulation, and metabolic status.
Dual Technology Platforms for Diverse Clinical Needs:
ekingst's EtCO₂ product line offers both Mainstream and Sidestream solutions to meet different clinical requirements.
Mainstream: The sensor connects directly to the airway interface without additional sampling tubing, offering the fastest response — ideal for adult and pediatric ICU and anesthesia scenarios, with minimal airway dead space and signal attenuation.
Sidestream: Gas is drawn through a fine sampling tube to the main unit for analysis. Since the sensor does not contact the airway, this configuration is well-suited for neonates, pediatric patients, and long-duration sleep monitoring — and integrates easily with portable ventilators and transport monitoring systems.
Core Technical Highlights:
Precise EtCO₂ Detection: Based on infrared absorption, the system delivers real-time end-tidal CO₂ measurement with high accuracy and minimal drift, providing a reliable foundation for clinical decision-making.
Real-Time Respiratory Rate Output: Alongside EtCO₂, the system continuously tracks spontaneous or mechanical ventilation rate, assisting in identifying respiratory events.
Professional Capnogram Waveform Analysis: Outputs standard CO₂ waveform curves (Phase I–IV), enabling morphology analysis to help detect circuit abnormalities (leaks, obstructions, misconnections), alveolar ventilation changes, and cardiopulmonary interactions.
PEEP Compatibility: When used in mechanically ventilated patients, the system tolerates a range of positive end-expiratory pressure, ensuring measurement accuracy independent of ventilation settings.
Typical Application Scenarios:
Anesthesia Depth Monitoring: EtCO₂ is a rapid, non-invasive method for confirming endotracheal tube placement and serves as a core parameter for continuous intraoperative ventilation monitoring, effectively preventing hypoventilation-related complications.
Critical Care Ventilation Management: In ICU patients on mechanical ventilation, EtCO₂ provides a reliable non-invasive estimate of arterial CO₂ partial pressure (PaCO₂), guiding ventilator parameter adjustments — particularly valuable in goal-directed ventilation strategies requiring strict PaCO₂ control.
Emergency and Transport: Quick connection enables immediate EtCO₂ readings, allowing rapid assessment of patient ventilation status in pre-hospital and in-hospital transport settings.
Sleep Respiratory Health: In sleep medicine centers and home screening settings, EtCO₂ monitoring is a key parameter for identifying obstructive sleep apnea (OSA), central sleep apnea (CSA), and hypopnea events. Combined with pulse oximetry (SpO₂), it enables comprehensive sleep respiratory monitoring.
4. International Quality Certification, Safeguarding Every Breath
The MDR CE certification confirms that ekingst's Multi-Gas Analysis and EtCO₂ monitoring product lines have achieved the highest EU regulatory standards across design and development, quality management, risk control, and clinical performance.
MDR (EU Medical Device Regulation 2017/745) is one of the most influential global medical device regulatory frameworks, with technical documentation requirements, clinical evidence standards, and post-market surveillance systems at the forefront of international best practices. MDR certification grants ekingst the regulatory clearance to legally market its respiratory monitoring products in the EU and globally — and provides international partners and end users with trustworthy quality assurance.
ekingst remains committed to advancing respiratory gas monitoring technology. With the mission of "Precision Monitoring, Safeguarding Every Breath," we will continue delivering safer, more reliable, and more accurate EtCO₂ monitoring and multi-gas analysis solutions to clinical professionals and patients worldwide.
